27days until NEET 2026Every question counts. ✊
⚗️
Did you know?
Diamonds and graphite are both made of pure carbon — just arranged differently.
Did you know?
Diamonds and graphite are both made of pure carbon — just arranged differently.
In which of the following compounds, the C—Cl bond ionization shall give the most stable carbonium ion?
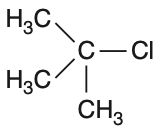
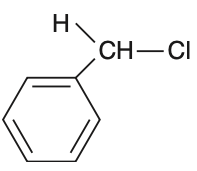
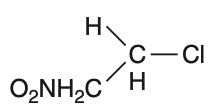
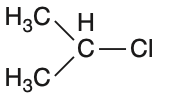
To determine which compound forms the most stable carbonium ion upon C—Cl bond ionization, we need to analyze the stability of the resulting carbocations.Carbocation stability is influenced by:• Hyperconjugation• Resonance• Inductive effectsLet's analyze each option:Option 1:
More practice, more score
Use hints to get start solving
Ask any question, get instant answers
Get detailed step by step solutions
Read while solving
Improve every day