27days until NEET 2026Every question counts. ✊
🫀
Did you know?
Your heart beats ~100,000 times a day — roughly 3 billion beats in a lifetime.
Did you know?
Your heart beats ~100,000 times a day — roughly 3 billion beats in a lifetime.
One mole of an ideal diatomic gas undergoes a transition from A to B along a path AB as shown in the figure. The change in internal energy of the gas during the transition is
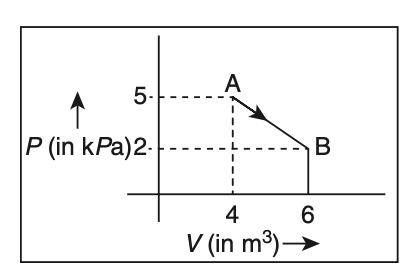
kJ
J
kJ
kJ
To find the change in internal energy of the gas, we use the formula:For a diatomic ideal gas,
More practice, more score
Use hints to get start solving
Ask any question, get instant answers
Get detailed step by step solutions
Read while solving
Improve every day