27days until NEET 2026Every question counts. ✊
⚗️
Did you know?
Diamonds and graphite are both made of pure carbon — just arranged differently.
Did you know?
Diamonds and graphite are both made of pure carbon — just arranged differently.
D-(+)-glucose reacts with hydroxylamine and yields an oxime. The structure of the oxime would be
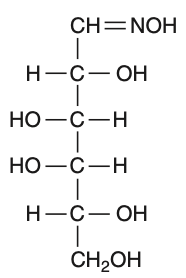
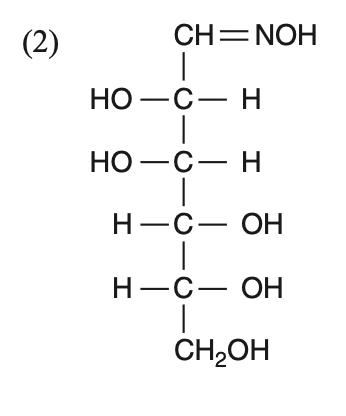
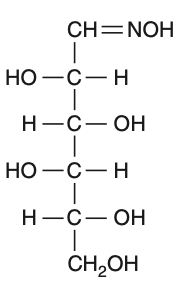
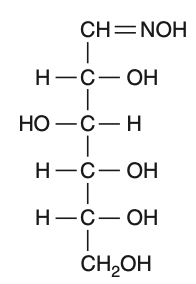
To solve this problem, we need to understand the reaction of D-(+)-glucose with hydroxylamine.• D-(+)-glucose is an aldohexose, meaning it contains an aldehyde group at the first carbon.• Hydroxylamine (NHOH) reacts with the aldehyde group to form an oxime.The reaction can be represented as:Aldehyde group:
More practice, more score
Use hints to get start solving
Ask any question, get instant answers
Get detailed step by step solutions
Read while solving
Improve every day