Did you know?
Absolute zero (−273.15°C) is the coldest possible temperature — at which atoms nearly stop moving.
Did you know?
Absolute zero (−273.15°C) is the coldest possible temperature — at which atoms nearly stop moving.
Which of the following p-V curves represents maximum work done?
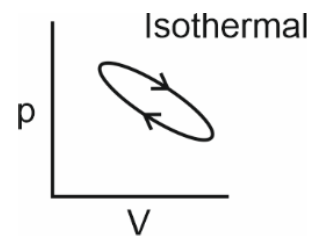
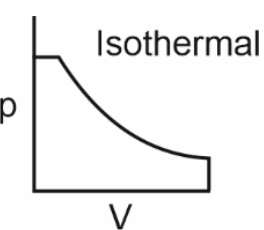
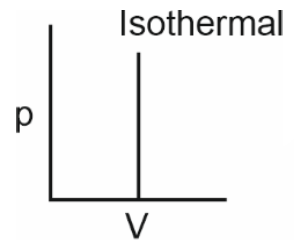
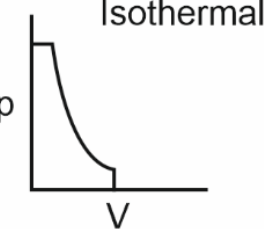
To determine which p-V curve represents maximum work done, we need to analyze the area under each curve.The work done by a gas during expansion or compression is given by the area under the p-V curve.Let's evaluate each option:• Option 1: The curve is a closed loop, indicating a cyclic process. The net work done is the area enclosed by the loop.• Option 2: The curve is a downward-sloping isothermal process. The area under this curve is significant, as it represents work done during expansion.• Option 3: The curve is a vertical line, indicating no change in volume. Hence, no work is done.
More practice, more score
Use hints to get start solving
Ask any question, get instant answers
Get detailed step by step solutions
Read while solving
Improve every day